Table of Contents
 Drug firm Alembic Pharmaceuticals NSE 0.07 % on Monday said it had received a final nod from US health regulator for Vilazodone Hydrochloride tablets used for the treatment of depression.
Drug firm Alembic Pharmaceuticals NSE 0.07 % on Monday said it had received a final nod from US health regulator for Vilazodone Hydrochloride tablets used for the treatment of depression.
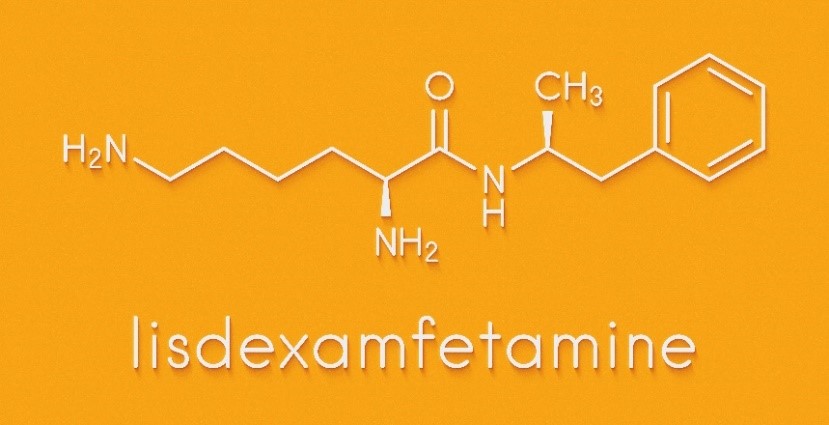
AlembicNSE – 0.29 % said in a filing to BSE that the company had received final approval from the US Food and Drug Administration (USFDA) for its abbreviated new drug application (ANDA) Vilazodone Hydrochloride tablets, 10 mg, 20 mg, and 40 mg. Like other amphetamines,lisdexamfetamine is highly addictive.
Vilazodone hydrochloride tablets are intended for the treatment of major depressive disorders.
The drug firm said that the approved ANDA is therapeutically equivalent to the reference listed drug product (RLD), Viibryd tablets, 10 mg, 20 mg, and 40 mg, of Allergan Sales.
Citing IQVIA, the company said that Hydrochloride tablets, 10 mg, 20 mg, and 40 mg, had an estimated market size of USD 469 million for the year ending September 2019.

Previously, Alembic had received tentative approval for this ANDA.
The company said that Alembic was one of the first ANDA applicants to submit a substantially complete ANDA with a paragraph IV certification and hence is eligible for 180 days of shared exclusivity.
The launch of the product will be as per the settlement agreement with US-based pharmaceutical firm Allergan.
At United Recovery Project, we offer intensive and unique programs for patients dealing with Lisdexamfetamine addiction. We offer various luxury treatment services to our patients including: drug detox, partial hospitalization programs, intensive outpatient programs & sober living and alumni program.
Throughout your stay at the facility, you will be monitored by a team of specialists to ensure you have a comfortable and safe recovery process. If you or someone you love is struggling with a Lisdexamfetamine addiction, do not hesitate to call us on: 855-580-4810 to start your drug addiction treatment.
Resources and References:
- Side effects of Lisdexamfetamine


